Promoting Partnerships To Improve Veterans’ Health
Promoting Partnerships To Improve Veterans’ Health
- Home
- About Our Members
- NPC Statutes
NPC Statute and VHA Handbook 1200.17
| NPC Statute: A revised NPC authorizing statute was signed into law on May 5, 2010. It was passed by Congress as title VIII of S. 1963 and became Public Law 111-163. It is codified at 38 USC §§7361-7366. NPC Authorizing Statute – 38 USC 7361-7366 – May 2010 [Word doc] NPC Authorizing Statute – 38 USC 7361-7366 – May 2010 [pdf] VHA Handbook: VA regulations specific to NPCs are contained in VHA Handbook 1200.17 (formerly M-3, Part I, Chapter 17). Handbook 1200.17 was revised and published in 2017. The Handbook is for research as well as research and education foundations. |
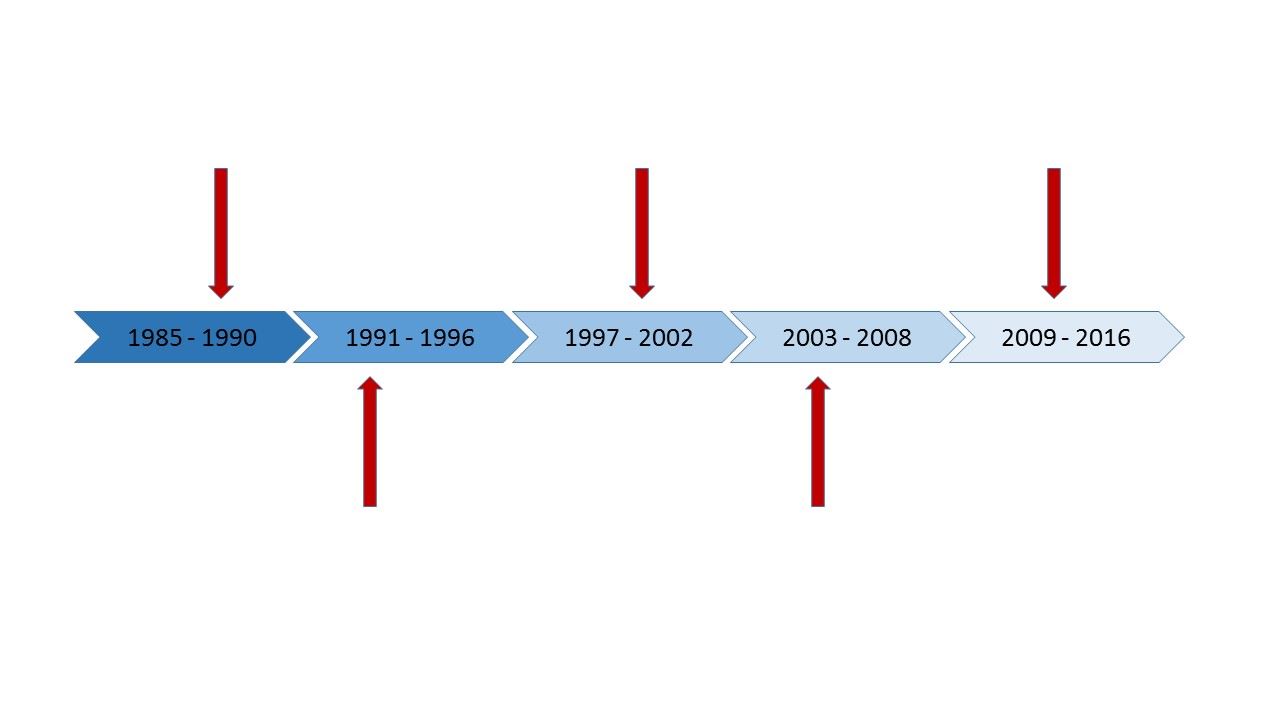
NPC Legislative Timeline